Advances in filter technology enable improved multiphoton microscopy performance.
Dr. Craig Hodgson and Dr. Turan Erdogan, Semrock Inc.
Multiphoton fluorescence microscopy
is similar to conventional fluorescence microscopy in that it images fluorescence
from molecules that tag a target of interest in a cell. However, in a two-photon
microscope, each fluorescent photon results not from a single-excitation photon,
but from two photons absorbed simultaneously — each with twice the wavelength
and half the energy of the equivalent single-excitation photon (Figure 1).
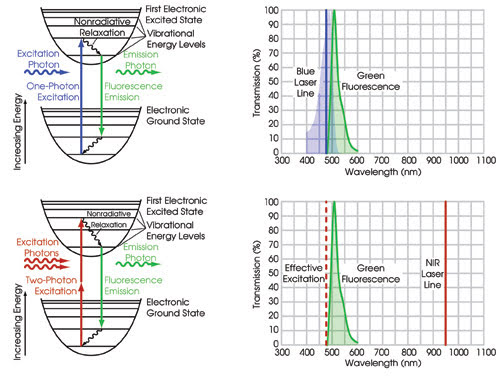
Figure 1. In standard single-photon fluorescence microscopy (upper
pair of images) a laser or filtered light source excites fluorescent molecules from
the ground state to the first excited state via absorption of a single photon for
each emitted photon. The most efficient excitation happens when the wavelength of
the excitation source matches the peak wavelength of the absorption curve, shown
as the blue shaded region. Because this occurs very close to the emission band (green
shaded region), one must sacrifice some of the desired fluorescence signal to minimize
the excitation light noise. In multiphoton fluorescence microscopy (bottom two images)
the same fluorescent molecule is excited with two photons (each twice the wavelength
and thus half the energy of the original one-photon excitation) instead of one.
The emitted fluorescence is essentially identical
to that which would have resulted from excitation of a single photon at half the
wavelength. Since the wavelength of the excitation source is now far removed from
the wavelength of the emitted fluorescence, one can use a filter that collects a
greater range of wavelengths over the emission band, yielding improved sensitivity.
It also is possible to do three-photon fluorescence, where three photons, each with
one-third the energy, are absorbed simultaneously to excite the molecule, replacing
the original one-photon excitation.
A typical multiphoton imaging system
comprises an excitation laser, scanning and imaging optics, a sensitive detector
(usually a photomultiplier tube), and optical filters, such as a dichroic beamsplitter
for separating the fluorescence from the laser and emission filters for blocking
undesired laser light from reaching the detector (Figure 2). A longer-wavelength
excitation source with sufficient peak intensity, such as a mode-locked, tunable
Ti:sapphire laser, is used because the two photons must arrive at the fluorescent
molecule at exactly the same instant to excite it.
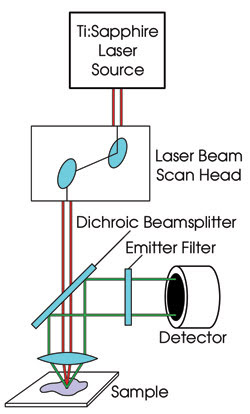
Figure 2. In a typical configuration of a multiphoton fluorescence
microscope, the high-peak-intensity (but moderate-average-intensity) pulsed laser
source is focused on the sample and raster scanned, just as in confocal microscopy.
The fluorescence is collected by a sensitive detector, typically a photomultiplier
tube, using a dichroic beamsplitter to separate the fluorescence from the laser
beam and an emitter to block the undesired laser light from the detector while transmitting
as much fluorescence as possible.
A multiphoton imaging system offers
many advantages. It provides true three-dimensional imaging, or optical sectioning,
as does confocal microscopy. The technique can image deep inside live tissue (Figure
3), eliminates undesired fluorescence from outside the focal plane, and reduces
photobleaching away from the focal plane, which increases sample longevity.
Multiphoton imaging allows imaging
of fluorescent dyes with very short Stokes shifts and/or very low efficiencies,
and even of inherently fluorescent molecules native to the sample or tissue. Its
growing popularity is based on the availability of modern high-peak-power pulsed
lasers, such as a widely tunable mode-locked Ti:sapphire. However, the technique
has traditionally suffered from the lack of commensurately high-performance optical
filters that provide excellent throughput across the whole emission range of interest
and sufficient blocking across the laser tuning range.
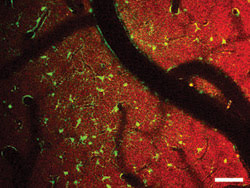
Figure 3. In
this two-color in vivo two-photon image from an exposed mouse cortex, NADH fluorescence
is shown as red and sulforhodamine-labeled astrocytes as green. Courtesy of Karl
A. Kasischke and Nikhil Mutyal, University of Rochester Medical Center.
Optical filters
Thanks to recent advances in optical filters,
multiphoton users now can achieve ultrahigh transmission in the filter passbands,
very steep transitions and deep blocking everywhere it is needed. These filters
represent a simple and — relative to the rest of the system — inexpensive
path to substantially boosting system performance.
The new breed of emission filters provides
clear transmission from the near-UV to the near-IR (Figure 4). In fact, the filters
look as clear as window glass, in contrast to the brownish tint of traditional filters.
And the new high-damage-threshold dichroic beamsplitters are designed to reflect
the precious fluorescence signal with exceptionally high efficiency. This throughput
advantage is of critical importance, because the multiphoton fluorescence process
is less efficient than conventional fluorescence; thus, it is desirable to collect
every possible emission photon.
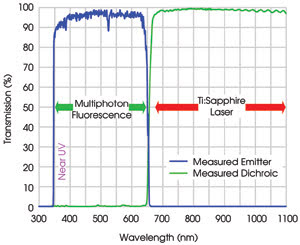
Figure 4. Typical measured spectra of
the near-UV and visible emitter FF01-680/SP and dichroic beamsplitter FF665-Di01
are displayed. This filter combination provides high throughput from the near-UV
(350 nm) through 650 nm, and very high OD >8 blocking along with high dichroic
transmission across the entire commonly used Ti:sapphire laser tuning range, including
the short-wavelength region down to 680 nm. It is ideal for near-UV fluorescence
imaging.
For best discrimination of the signal
from scattered laser background noise, the emission filters also must provide deep
blocking across the entire Ti:sapphire laser tuning range. Because of their sensitivity,
photomultiplier tubes are the most common means of detecting multiphoton emission,
but they also are sensitive to the excitation light, especially at the short end
of the laser tuning range. Thus, high blocking is critical to achieve a high signal-to-noise
ratio and measurement sensitivity. Without sufficient filter blocking, it is not
uncommon for a substantial fraction of the measured signal (even tens of one percent)
to consist of scattered laser light. This noise source can be almost completely
eliminated with the new multiphoton filters.
We have found that when using these
filters in a typical experimental setup, the signal collection efficiency
increased by 56 percent and at the same time the increased blocking of stray excitation
light led to a 44 percent decrease in background noise — as a result, the
signal-to-background ratio increased from about 7 to over 20. These numbers represent
a significant improvement for this emerging imaging technology.
Sometimes it is desirable to restrict
the spectral band of fluorescence emission that is detected, especially when multiple
fluorophores are used to label different targets in a sample. Narrower bandpass
emission filters are ideal for this purpose. Because standard bandpass fluorescence
filters often do not provide sufficient blocking of a multiphoton excitation laser,
researchers have typically had to combine multiple filters to achieve acceptable
signal-to-noise performance. With the exceptional transmission of the new breed
of multiphoton filters, researchers now can leave these filters fixed in their system
to block the laser, and then exchange narrower bandpass emission filters when necessary
to limit the fluorescence emission band for multi-fluorophore work.
Peering deeply
Dr. Karl A. Kasischke and his co-workers at the
Center for Aging and Developmental Biology in the University of Rochester Medical
Center’s department of neurosurgery in Rochester, N.Y., use multiphoton fluorescence
microscopy to measure the intrinsic fluorescence from nicotinamide adenine dinucleotide
(NADH) as a functional imaging signal in the intact and living brain. NADH plays
a crucial role in energy metabolism in the brain and is a fluorescent intracellular
molecule, so it is possible to use multiphoton fluorescence microscopy to directly
measure the concentration and location of this important coenzyme without introducing
foreign dyes or tracers.
Importantly, the reduced co-enzyme
(NADH) is fluorescent, while the oxidized co-enzyme (NAD+) is not. Therefore, intrinsic
NADH fluorescence yields a direct measure of the cellular NADH/NAD+ ratio and has
been used as an indicator for both oxidative (production of ATP) and glycolytic
(anaerobic glucose metabolism) metabolism.
Kasischke and his colleagues have previously
shown that two-photon imaging and spectroscopy of NADH provide quantitative and
subcellular imaging of metabolic transitions in brain tissue. The in vivo application
of this imaging technology offers great potential for the cellular localization
of brain activation and for the investigation of the underlying biochemical changes.
In another example, Edward Brown III
and his co-workers in the department of biomedical engineering at the University
of Rochester Medical Center use multiphoton fluorescence microscopy to image deeply
into internal regions of tissue and tumors. Intravital microscopy coupled with chronic
animal window models has provided stunning insight into tumor pathophysiology, including:
gene expression; angiogenesis; cell adhesion and migration; vascular, interstitial
and lymphatic transport; metabolic microenvironment; and drug delivery. However,
prior to the use of multiphoton fluorescence microscopy, analysis was limited to
the tumor surface (<150 μm deep). Multiphoton microscopy can provide high-resolution,
three-dimensional images of gene expression and function in deeper regions of tumors.
These insights could be critical to the development of novel therapeutics that target
not only the tumor surface, but also internal regions (Figure 5).
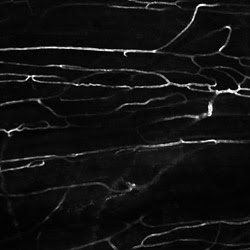
Figure 5. This in vivo two-photon image
shows blood vessels in the dorsal skin of a live mouse labeled with intravenous
injection of FITC-Dextran. Courtesy of Edward Brown, University of Rochester Medical
Center.
The researchers also are using a closely
related imaging technique to study collagen in tumors. Called second-harmonic-generation
microscopy, it involves a nonabsorptive conversion of two long-wavelength photons
into one short-wavelength photon of the same total energy. The emitted photon results
from a nonlinear optical interaction and is distinct from a fluorescent photon,
which always has a lower energy than that of the excitation photon or photons.
In tumors, fibrillar collagen is a
significant source of second-harmonic generation. The content and structure of collagen
are essential for governing the delivery of therapeutic molecules to tumors. By
using second-harmonic generation, Brown and his co-workers imaged fibrillar collagen
within tumors growing in mice. Using this noninvasive technique, they quantified
the dynamics of collagen modification after pharmacologic intervention and provided
mechanistic insight into improved diffusive transport induced by the hormone relaxin.
This technology could enhance scientists’
and clinicians’ ability to estimate the relative penetrabilities of molecular
therapeutics. And the new optical filters play a vital role in supplying second-harmonic-generation
microscopy with the sensitivity needed for these applications.
The technology
Optical filters are constructed by depositing
alternating thin-film layers of high and low index of refraction. Typically, each
layer has an optical thickness equal to roughly one-fourth of the wavelength of
light, where optical thickness is the product of a layer’s real thickness
and its index of refraction. For example, green light has a wavelength of 0.5 μm
(millionths of a meter), so the corresponding quarter-wave layer thickness is about
50 to 80 nm (billionths of a meter). Traditionally, thin-film filters have been
constructed with tens of layers, and they have required many coatings for a single
complex filter such as a multiphoton emission type. The new generation of high-performance
filters is constructed with many hundreds of layers in each coating, so that each
layer’s thickness must be controlled with utmost precision. To achieve optimal
spectral performance, sophisticated control of the thin-film deposition process
is required to compensate for layer thickness errors.
The technology that meets these requirements
is based on a combination of ion-beam-sputtering deposition systems (long renowned
as the premier coating technology for low-loss, high-performance mirrors and filters)
with an advanced deposition control technology. These filters are based on refractory
metal-oxide materials that are as hard as the glass on which they are coated, with
a single glass substrate design coated on one or both sides. There are no adhesives
or epoxies in the optical path, eliminating the filter degradation, undesirable
loss and autofluorescence that contribute to increased background.
The hard-coating technology and all-glass
design of the new breed of filters provide exceptional reliability: The filters
can be cleaned and handled like any standard glass optics, they have a laser-damage
threshold higher than the laser power used in multiphoton microscopy, they do not
photodarken or “burn out,” and they are impervious to environmental
factors such as temperature and humidity extremes.
Multiphoton microscopy is a vital biological
imaging technique, and recent advances in optical filters are making it possible
for researchers to significantly upgrade their systems’ performance. Continued
improvements in technology, especially in the excitation lasers, will further enhance
the technique’s efficiency and widescale accessibility.
Meet the authors
Craig W. Hodgson is a principal engineer and Turan
Erdogan is the chief technology officer at Semrock Inc., Rochester, N.Y.; e-mail:
[email protected].