Drug discovery efforts may get a boost from an imaging technique based on superresolution microscopy and fluorescence correlation spectroscopy.
Called fcsSOFI — for fluorescence correlation spectroscopy superresolution optical fluctuation imaging — the technique allows researchers to observe the nanoscale spaces inside porous materials. These materials are used in chromatography, a process through which proteins are separated and purified depending on the different rates at which they flow through the pores.
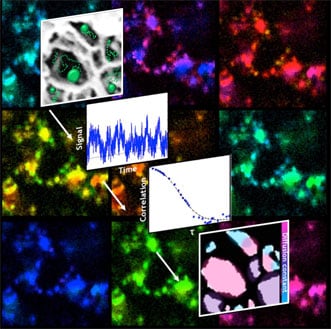
Fluorescent particles diffuse through a porous nanostructure, revealing the arrangement of the pores. Courtesy of the Landes Research Group/Rice University.
While fluorescent tags are commonly used to image structures in biology and elsewhere, they typically haven't been used to explore empty spaces.
"That's the problem we had to solve to understand what was going on in the separation material," said Christy Landes, associate professor of chemistry and electrical and computer engineering at Rice University and leader of the study.
Imaging porous materials with atomic force, x-ray and electron microscopy would require the samples to be either frozen or dried, which would change their structures, Landes said. On the other hand, observing the fluctuations of fluorescent tags as they flow through the samples using fcsSOFI allows nondestructive measurement of pore dimensions and dynamics.
The lab tested its technique on both soft agarose hydrogels and lyotropic liquid crystals, finding it improved pore resolution by up to a factor of 2, compared with diffraction-limited fluorescence imaging and single-particle tracking.
 "We now have both pieces of our puzzle," Landes said. "We can see our proteins interacting with charges within our porous material and we can measure the pores. This has direct relevance to the protein separation problem for the $100 billion pharmaceutical industry."
"We now have both pieces of our puzzle," Landes said. "We can see our proteins interacting with charges within our porous material and we can measure the pores. This has direct relevance to the protein separation problem for the $100 billion pharmaceutical industry."
Next the researchers plan to extend their mapping to 3D spaces. The fcsSOFI technique could benefit the analysis of porous materials of all kinds, they said, including liquid crystals, hydrogels, polymers and even biological substances such as cytosol, the compartmentalized fluids in cells.
Researchers from the University of California, Los Angeles and Kansas State University also took part in the project. Funding came from the Welch Foundation, National Science Foundation and Department of Energy.
The findings were published in ACS Nano (doi: 10.1021/acsnano.5b03430).