In order for biologists, drug developers, clinical lab professionals and other scientists to fully exploit new microscopy techniques and applications, parallel developments in laser technology are often required. Laser manufacturers are responding with products optimized to match the needs of these applications.
Marco Arrigoni, Nigel Gallaher, Darryl McCoy, Volker Pfeufer, Matthias Schulze, and Daniel Callen, Coherent Inc.
Laser development for the microscopy market continues to be driven by key trends in applications, which currently include superresolution techniques, multiphoton applications in optogenetics and other areas of neuroscience, and even a shift in multiphoton imaging toward preclinical and clinical usage.
In spite of its long history, optical microscopy — particularly laser-based — is a very dynamic field. New techniques continue to be developed, while existing techniques are being applied to new applications.
Superresolution microscopy — optical switching of fluorescent labels
In order to better understand the details of processes like signaling and the control of gene expression, biologists need to correlate molecular-level events with macroscopic structures and dynamics. This has fueled rapid growth in superresolution microscopy techniques, often referred to as nanoscopy, that go beyond the spatial resolution limit set by diffraction. This limit is about half the wavelength of light (i.e., for visible light, approximately 200 to 250 nm in the XY plane) and, in the case of confocal microscopy, about 500 nm in the Z direction. Most superresolution techniques use CW lasers, often with fast internal or external (on/off) modulation.
All optical superresolution or nanoscopy techniques are based on the principle of optically and reversibly preparing states of a fluorescence label that differ in their emission characteristics (e.g., a bright “on” and a dark “off” state). Based on their different mechanisms for this on/off photoswitching, optical nanoscopy methods can be loosely divided into two groups: those that directly improve microscope effective spatial resolution by deterministic photoswitching in space and time, and those that achieve the higher resolution by (random) stochastically switching single-molecule fluorescence on and off in space.
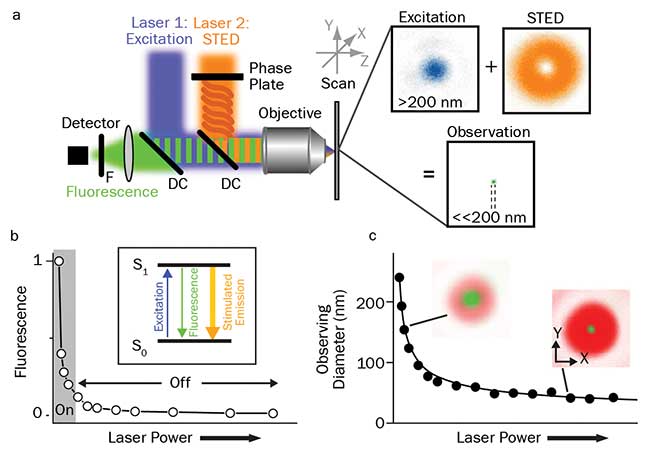
Figure 1. Principles of STED. Schematic drawing of the setup of a STED nanoscope with phase plate, objective lens dichroic mirror, fluorescence filter, detector, scanning device, and excitation and STED lasers with their focal intensity distribution (right), and a representative, subdiffraction-sized observation area (a). STED nanoscopy is based on inhibiting fluorescence emission by de-exciting the excited S1 “on” state to the S0 “off” ground state via stimulated emission. Increasing the power of the STED laser drives the inhibition into saturation (b). Combined with an intensity distribution that features at least one intensity zero, this on/off fluorescence inhibition realizes subdiffraction-sized observation volumes — the volume in which fluorescence emission is still allowed (green insets) decreases with increasing STED laser power (c). Courtesy of Christian Eggeling.
Examples of the former include stimulated emission depletion (STED) microscopy (Figures 1 and 2), ground state depletion microscopy, reversible saturable optical fluorescence transition (RESOLFT) microscopy or parallelized RESOLFT microscopy (often denoted nonlinear, or saturated structured illumination microscopy (SIM)). Examples of the latter include direct stochastic optical reconstruction microscopy (d-STORM) and fluorescence photoactivation localization microscopy (f-PALM).
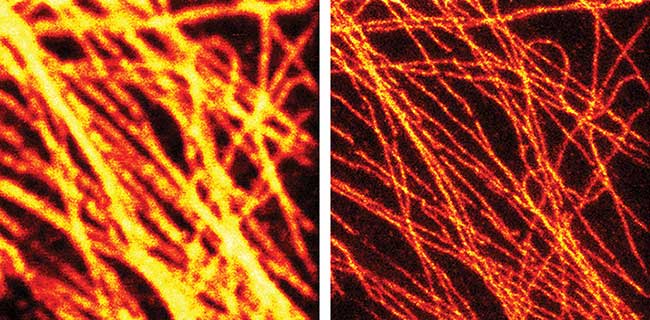
Figure 2. The effectiveness of STED is clear in this pair of images of fluorescently tagged microtubuli in mammalian cells using a 577-nm laser to drive the STED effect: conventional confocal (left) and gated STED image (right). Courtesy of Dr. Giuseppe Vicidomini and Prof. Alberto Diaspro, Italian Institute of Technology.
Stochastic-based techniques such as f-PALM and d-STORM also use two lasers (Figure 3). Here, one laser is used to drive most of the fluorophores between the “on” and “off” states, so that only a very small, random subset of well-separated molecules are in their “on” state and available to be excited by a second laser. The fluorescence spread from each of these camera-imaged point sources is then analyzed to find the centroid (the point source or spatial localization, based on the point spread function, or PSF, of the microscope). Repetitive on/off switching brings up different random subsets in subsequent camera images for single-molecule localization. After many cycles, the microscope computer assembles all of the idealized point sources into a subdiffraction image, since the localization of a single molecule can be performed with high subdiffraction spatial precision.
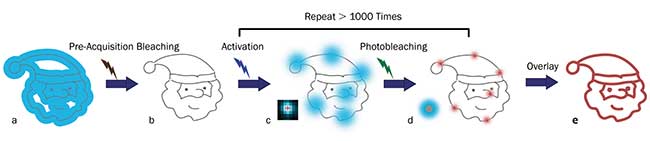
Figure 3. Principles of STORM microscopy. A wide-field fluorescence image (blue area) is observed from overlapped fluorophores in a small target structure (gray lines) (a). Little to no fluorescence is left after pre-acquisition bleaching with an appropriate laser (b). Individual fluorophores are sparsely activated by an activation laser, and then emit fluorescence (blue spots) after applying an excitation laser (usually the same one for pre-acquisition bleaching) (c). The excitation laser is again used to bleach the fluorophores. The center positions (red crosses) of the fluorophores are found using an appropriate molecule localization algorithm. The uncertainty of the center positions (red spots) is significantly smaller than the diameter of the single molecule fluorescence from the same fluorophores (large blue spot in the left corner) (d). Repeating the activation, excitation, localization, and photobleaching process more than 1000 times allows reconstruction (via overlaying the center positions of the fluorophores) of a superresolution image of the target structure (red lines) (e). Courtesy of Zhen-li Huang, Wuhan National Laboratory for Optoelectronics, Huazhong University of Science and Technology.
Trends in CW lasers to support superresolution techniques
Laser manufacturers are supporting the rapid growth in superresolution microscopy with new products that provide higher and easily adjustable power, new wavelengths, and simplified fiber coupling and fiber combining.
Higher laser power is necessary because researchers want output in the 1- to 3-W range for STED, and typically a few hundred milliwatts for f-PALM and d-STORM. And in the case of SIM, microscope manufacturers are looking to move up to 300-mW lasers to decrease image acquisition times, whereas early users of this method worked with as little as 100 mW. Both deterministic and stochastic fluorescence switching require adjustable power output in the “bleaching” laser in order to optimize these techniques.
Lasers based on optically pumped semiconductor laser (OPSL) technology are proving ideal to meet these higher/adjustable power needs, as OPSL technology is readily scalable simply by increasing the pump diode power. And the OPSL output can be smoothly adjusted from near zero to maximum power with no effect on beam quality or beam pointing; this technology does not suffer from thermal lensing, which prevents wide power adjustment in diode-pumped solid-state (DPSS) and other solid-state lasers.
Expanded range of CW laser wavelengths
Another key trend is the growing demand for new laser wavelengths particularly at the far end of the visible spectrum to excite many of the new fluorophores used in microscopy. These include the mFruit series of fluorescent proteins and a host of new green, yellow, and orange excited dyes originally developed for multichannel flow cytometry applications. And, as with wide-field microscopy, new nanoscopy methods often use combinations of multiple fluorophores to permit simultaneous mapping or probing for more than one type of structure or biochemical.
Wavelength scalability is a key advantage of OPSL technology, compared to older laser technologies with inherently fixed wavelength properties. The OPSL can be fabricated for any arbitrary output wavelength across much of the visible and near-UV spectrum merely by fine-tuning the design of the gain semiconductor chip. This has led to a paradigm shift in laser applications development. Traditionally, a new fluorophore would be excited with the closest available laser wavelength; indeed many early fluorophores were developed to match the 488 nm argon ion wavelength. With OPSL technology, the situation reverses and the laser can optimally match the new fluorophore and/or application.
For example, lasers at 588 nm and 594 nm were specifically developed for exciting orange fluorophores. Other models at 550 nm have also been developed, as this wavelength can be used to simultaneously excite green dyes and several new yellow fluorophores with a single laser. Another relatively new OPSL wavelength is the legacy (krypton ion) wavelength of 568 nm.
Laser diodes and OPSLs are increasingly complementary technologies that are now both offered in common plug-and-play packages. In addition to the new wavelengths based on OPSL technology, several other new wavelengths utilize laser diode technology.
An example is at 980 nm for microscopy relying on upconversion of nanoparticles (UCNP). This is a method that addresses the wavelength trade-off in deeper imaging with single-photon microscopy. Longer wavelengths enable deeper penetration (less scatter) but red (Stokes) shifted fluorescence leads to low quantum efficiency and hence low signal-to-noise by silicon-based cameras and detectors. With UCNP, the laser excites the sample in the 980-nm region, and the emission is blue-shifted, peaking at 800 nm. In 2017, a new laser diode was developed providing up to 150-mW of output at 980 nm to optimally support UCNP imaging.
Looking forward, a growing number of fluorophores need excitation light in the 750- to 755-nm window. Standout examples include Dylight 755nm, Alexa 750, iFluor 750-streptavidin conjugate, IRDye 750 Infrared Dye, and Lumenprobe Cy7. Expect to see new lasers at 752 nm in 2018 to optimally excite these fluorophores.
Neuroscience dominates growth in multiphoton imaging
Multiphoton excitation (MPE) microscopy relies on the uniquely high peak power and short pulse duration of ultrafast lasers; the infrared laser light penetrates more deeply than visible light and is absorbed only at the focused beam waist, providing natural sectioning capability. In MPE, the single biggest driver in techniques and laser development is neuroscience. Billions of dollars in public and private funding are enabling dramatic advances in understanding how the brain processes information, from the individual synapses to the level of the neural network. Moreover, increased life expectancy brings an emphasis on improved quality of life in old age, and other neuroscience goals include a better understanding of how to reduce and treat age-related mental impairments such as Alzheimer’s and Parkinson’s. MPE is playing a key role in much of this research because it enables deep (1- to 1.5-mm) in vivo imaging with full 3D resolution and low photodamage. The classic mammalian setup for neuroscience studies involves a mouse with a thin glass cover replacing a small part of the skull to provide visual access to the cortex at layers 1 through 6. Other MPE applications include cancer research, stem cell studies, and other aspects of immunology. While many of these applications involve laboratory research, there is also growing interest in the elusive goal of moving toward clinical (i.e., human) applications.
The overarching trends in neuroscience rely on faster and deeper in vivo imaging. The reason is that researchers increasingly want to simultaneously study as many neurons as possible to understand how their collective activity enables brain functions. At the same time, novel applications are being enabled by new fluorescent tools and methods, many of which use gene expressions in laboratory animals, from insects to mice. Advances in these genetically expressed probes include the development of more efficient red-shifted fluorescent probes for deeper imaging with less scatter, as well as new types of so-called functional probes. These functional probes include proteins whose fluorescent characteristics are modified by specific local physiological conditions that take place, such as pH and Ca2+ concentration.
Important advances in the field of neuroscience include the use of optogenetics, a technique pioneered by Dr. Karl Deisseroth at Stanford University and Edward Boyden at MIT who were among the recipients of the 2016 Breakthrough Prize in Life Sciences for their work.
Optogenetics uses a class of proteins called opsins that reside in the cell membrane and act as single-element gates for specific ions that can be controlled by pulses of light. When irradiated at the appropriate wavelength, opsins enable Na+, K+, or Cl- or other charged ions to flow across the membrane, leading to changes in the electric membrane potential. Neurons transmit signals along their length in the form of voltage spikes propagating along axons and dendrites. Thus, optogenetics allows researchers to use light to control the activity of target neurons by enabling or inhibiting the flow of these ions. Researchers use different light sources depending on the type of experiment. But the high spatial resolution and three-dimensional discrimination inherent to multiphoton excitation with ultrafast lasers make it the only current method that can be used to photoactivate or silence activity with single-neuron resolution.
Optogenetics can be combined in various ways with other experimental tools, e.g., patch clamp electrodes to monitor electrical activity of neurons. But one of the most sophisticated and powerful types of experiments is called all-optical physiology where ultrafast laser light is used to map the neuron anatomy, to perform activation and/or silencing, and to monitor resulting activity in connected neurons by using fluorescent probes able to map spatiotemporal changes in Ca2+ concentration to indicate neurotransmission activity. While synthetic calcium indicators have been used for decades, the development of so-called GECI, or genetically encoded calcium indicators, enables researchers to breed colonies of animals where specific types of neurons fluoresce more or less depending on Ca2+ concentration.
In all-optical physiology, it is important to avoid crosstalk between optogenetics activation and Ca2+ detection. A simple method of achieving this goal is to use probes with spectral responses to laser light well separated in wavelength (Figure 4); the range of different probes and opsins continues to be expanded in order to better facilitate this separation.
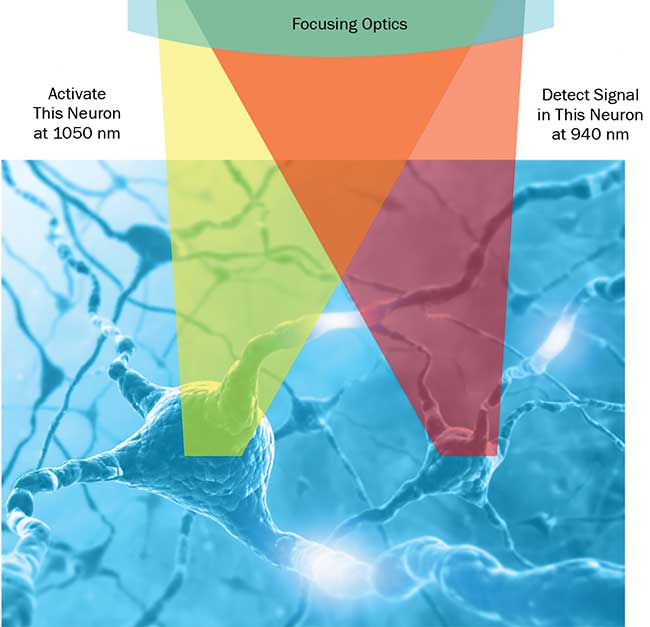
Figure 4. Advanced optogenetics. In all-optical physiology experiments with multiphoton lasers, neural activity can be manipulated and monitored at single-neuron resolution. The use of two separate wavelengths for photoactivation and detection enables minimization of crosstalk between these “write & read” channels. Courtesy of Coherent Inc.
All-optical physiology is not the only area of neuroscience where multiple ultrafast laser wavelengths are needed simultaneously. For example, an interesting technique for mapping different neuron types is “brainbow” imaging. Here, subject animals are transfected with genes for three or more different fluorescent proteins, whose emissions peak at red, green and blue wavelengths. The various cell types in the brain express these proteins in unique stochastic ratios, each characterized by a different ratio of the fluorophores and, therefore, a unique emission spectrum and color signal. Brainbow imaging needs two or more independently tunable ultrafast sources. CARS and SRS imaging are other techniques that similarly rely on two simultaneous ultrafast wavelengths.
Trends in ultrafast lasers driven by neuroscience needs
The overarching trends in ultrafast lasers for MPE are the use of longer wavelengths, higher power, and dual wavelength output.
Longer wavelengths are important for several reasons. Figure 5 shows the combined effects of scattering and other attenuation mechanisms (green solid line). The attenuation length increases monotonically as the wavelength increases from 800 nm to about 1300 nm, then drops due to water absorption but peaks again in the region between 1700 and 1800 nm. This trend fostered the development of probes — like fluorescent proteins, GECI and opsins — excitable in two-photon modality at wavelengths in excess of 1 µm. (This expanded palette also provides a useful tool for minimizing crosstalk in all-optical experiments.) Another benefit of longer wavelengths is the reduction of light-related damage to tissues under observation.
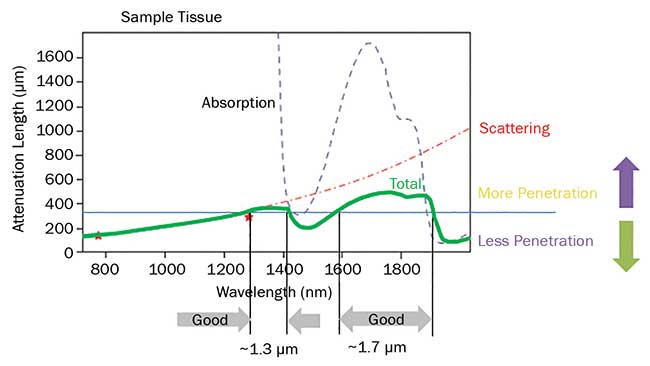
Figure 5. The penetration depth for ultrafast lasers in live brain tissue is limited by a combination of scattering and absorption. There are two optimum penetration windows centered at 1300 and 1700 nm. Based on a private communication with professor Chris Xy, Cornell University.
Higher power is advantageous for two reasons. First, it enables brighter images at greater depths within live brain tissue. Second, it enables the growing number of physiology studies that involve larger numbers of individual neurons. Neuronal activity occurs on the millisecond time-scale and, as a result, researchers are developing faster techniques to monitor or activate many neurons at the same time on this millisecond timescale, something unachievable by conventional raster scanning. The most common and flexible approach is to use a programmable spatial light modulator (SLM) as a phase mask to generate arbitrary patterns of laser spots in the sample. The SLM is used in different ways. For optogenetic activation of many neurons, the SLM is programmed to produce in parallel a diffraction-limited spot on each targeted neuron while a pair of scanners move this spot on each neuron until an action potential is triggered. Another approach is to create a spot matching the neuron size (about 10 µm) and either activate the neuron or measure its electrical activity (Ca2+) by illuminating all of it at the same time. Incidentally, this change in XY resolution would cause an unwanted and undesirable loss in Z axis resolution. Fortunately, this loss can be recovered by the so-called temporal focusing technique.
Until 2012, the only proven gain material for ultrafast lasers was Titanium:sapphire (Ti:S), whose tunable output power dies at 1080 nm. Longer wavelengths (up to 1600 nm) are then generated by using part or all of the Ti:S output to pump a tunable optical parametric oscillator (OPO). But the aforementioned experimental trends create a need for higher (>1 W) power at wavelengths greater than 1050 nm. These needs are unfulfilled by Ti:S-based systems. Because of this limitation, laser manufacturers turned to a new ultrafast gain material: ytterbium-doped crystal and fiber media. These materials are capable of very high power over a limited wavelength range — between 1030 and 1070 nm depending on the particular host composition. At first, it was not possible to generate both high (multiwatt) power and short (<150 fs) pulse width from ytterbium-based media, but engineers addressed this limitation in a number of ytterbium-based lasers such as the Chameleon Fidelity and Discovery. In the Discovery, an OPO is integrated with the ytterbium pump laser in the head to provide broad tuning to 1300 nm. Just as important, some of the fixed-wavelength ytterbium output can be used directly as the second wavelength in dual-wavelength experiments. This type of laser has now become the de facto standard for many MPE applications, not just those involving optogenetics.
However, neuroscience is an extraordinarily dynamic field and so it is not surprising that numerous researchers are requesting even higher power. One reason is the need to split the power to cover an even higher number of neurons. A state-of-the-art experiment might today involve a few tens of neurons. But ultimately researchers would like to simultaneously survey as many as 10,000 neurons, as in the case of a column of cortex measuring 250 × 250 μm with a depth up to 1 mm.
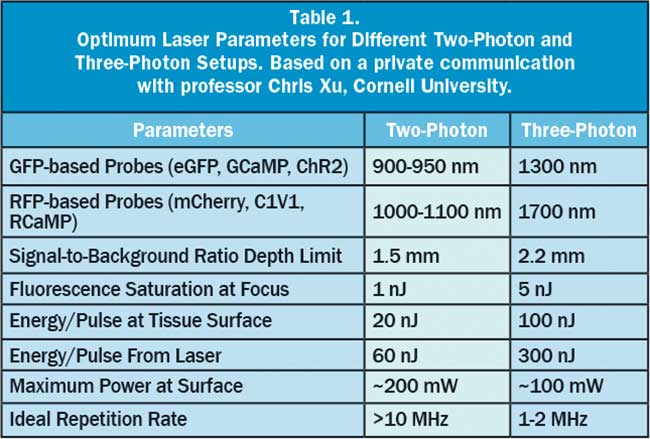
Another factor driving the demand for even higher power is the growing interest in using three-photon excitation in neuroscience, as this technique is proven to provide even deeper imaging capabilities than two-photon microscopy. Referring again to Figure 5, the light penetration in the model tissue has relative maxima around 1300 and 1700 nm. These wavelengths are too long for two-photon excitation of today’s fluorescent probes and optogenetics tools, but are ideal for three-photon excitation of probes used with 900- to 950-nm light when excited in two-photon regime, like GFP and GCaMP. The first experiments in three-photon functional imaging in fact used GCaMP excited at 1300 nm. Even more recently, red-shifted probes like tdTomato have been imaged using 1700 nm, which probably represents the longest usable wavelength for deep imaging of tissues. An enabling benefit of three-photon imaging comes from the high nonlinearity of this process, as very little fluorescence is excited away from the focal plane — much less than with two-photon. Consequently, the signal-to-background ratio is much better than for two-photon excitation, and imaging as deep as 1.6 mm has been demonstrated. In three-photon excitation, the highly nonlinear excitation mechanism requires very high peak power and pulse energy while the average power should remain below the thermal damage threshold, same as with two-photon excitation. As noted earlier, ytterbium — especially in fiber format — is very power scalable and flexible so that it is possible to increase the energy per pulse from the nanojoule to the microjoule level and to pump a wavelength conversion device like an OPA to access these longer wavelengths. The average power cannot, however, be increased ad infinitum, as it would result in thermal tissue damage. Instead there is a trade-off between pulse energy, repetition rate and average power. Table 1 summarizes typical optimum values for two-photon and three-photon excitation. To avoid tissue damage in three-photon methods, the average power is maintained at a moderate level by reducing the pulse repetition rate for these high-energy pulses to 1 MHz or less, compared with the 80 MHz of lasers for two-photon excitation.
One other applications-driven trend in ultrafast lasers for microscopy merits a brief mention here — laser reliability. The competition for funding in dynamic fields like neuroscience is greater than ever. This, in turn, creates pressure on researchers to produce better data and publish more papers. Productivity is thus recognized today as being as important as performance in ultrafast lasers. Manufacturers have responded to this challenge through improved reliability. A key part of this program has been the laser industry’s first use of rigorous HALT/HASS design and testing methods, where rapid temperature changes and vibration randomized in direction and frequency are applied to laser subsystems and to the final product as well. HALT stands for “highly accelerated life testing” and HASS stands for “highly accelerated stress screening.” These methods use third-party-accredited equipment and protocols to systematically identify and eliminate every potential fault, long before each laser ever reaches the customer.
Summary
Laser-based microscopy is one of the most valuable tools in the life sciences. As researchers develop and refine new techniques and methods, laser sources need to be adapted and re-optimized in order to fully exploit the potential of these techniques. By closely working with key researchers and institutions, laser manufacturers are responding in a timely manner with new products designed specifically for the changing needs of these applications.
Acknowledgments
The authors wish to acknowledge the assistance of Dr. Christian Eggeling (principal investigator, Human Immunology Unit, and scientific director, Wolfson Imaging Centre, Weatherall Institute of Molecular Medicine, University of Oxford, U.K.) in the form of personal communications about optical nanoscopy techniques and specifically STED technology, including supplying Figure 1. They also wish to acknowledge the assistance of professor Zhen-li Huang at Wuhan National Laboratory for Optoelectronics, Huazhong University of Science and Technology, for important insights in STORM imaging and for providing Figure 3. In addition, the authors thank professor Chris Xu of Cornell University for helpful discussions and the information used to create Table 1.